|
Overview of Cochlear Implants and other Implantable Hearing Devices
Introduction:
Hearing difficulty is a common presenting complaint and may be due to a number of causes. A conductive hearing loss occurs when sound energy cannot reach the inner ear, for example due to occlusion of the ear canal, perforation of the tympanic membrane, middle ear pathology such as fluid or erosion or fixation of the ossicles in the middle ear. A sensorineural hearing loss occurs when there is damage to the inner ear structures or the auditory nerve, for example due to infection, ischaemia or toxic drugs. Of course, the two types of hearing loss can occur together leading to a mixed loss. Some forms of conductive loss can be treated surgically, for example insertion of a ventilation tube for a middle ear effusion or ossiculoplasty for ossicular problems. Alternatively, a hearing aid can improve hearing in many forms of conductive loss. For sensorineural losses, we cannot currently repair damaged inner ear structures medically or surgically, so the only option is to increase the sound energy reaching the inner ear with a hearing aid. Recently, a number of implantable hearing devices have become available and this article aims to increase awareness of these devices.
Bone Conducting Hearing Devices (including BAHAs):
The bones of the skull are capable of transmitting sound energy to the inner ear, and this is the basis of bone conducting hearing aids and tuning fork tests. However, a traditional bone conducting aid can be uncomfortable, as it has to be pressed against the skull, and is inefficient, as transmission through scalp skin results in significant loss of energy. A bone anchored hearing aid circumvents these problems by implanting a titanium fixture and attaching a hearing aid to the fixture (Fig 1). Sound energy is amplified by the hearing aid and transmitted to the inner ear via the fixture and skull bones, bypassing the conductive loss. As there is little attenuation of energy across the skull, the contralateral cochlea is also stimulated. The choice of material is important. Titanium is particularly inert, and provided surgery is performed carefully to prevent damage to osteocytes, the titanium fixture osseointegrates with bone to provide an exceptionally strong bond, and this transmits acoustic energy with minimal wastage.
Fig1.Bone Anchored Hearing Aid
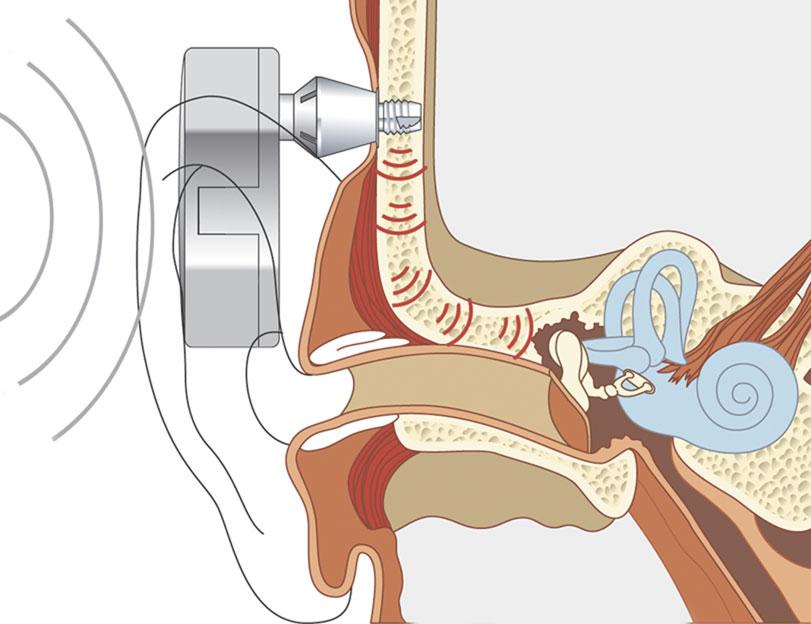
Indications: BAHAs are indicated for a mild to moderate conductive or mixed hearing loss not helped by conventional aids, including conventional bone conducting hearing aids. Cochlear function should be better than 45dB if a standard behind the ear BAHA is to be used, while body worn BAHAs can be used for hearing losses up to 60dB.
Children with congenital external or middle ear pathology but normal cochleas are good candidates for a BAHA, particularly if the problem is bilateral. A prosthetic auricle can be attached at the same time. Alternatively, reconstruction techniques can be considered although these are usually complex, require multiple operations and produce variable results.
Patients with chronically discharging ears, for example those with chronic suppurative otitis media or who have had mastoid surgery, and need a hearing aid, often find the hearing aid makes the ear sweaty and prone to infections and discharge. Surgical correction may not always be possible and BAHAs work well in these situations.
BAHAs produce good results in patients with otosclerosis and are increasingly being offered as a relatively safe alternative to stapedectomy.
In some cases surgery to improve hearing is possible but if the ear is the only hearing ear, patients may prefer to avoid the potential risk to hearing from middle ear surgery.
BAHAs are now also being marketed for unilateral hearing loss, for example after acoustic neuromas surgery. Conventionally, a CROS aid (Contralateral Routing of Signal) is used where a microphone positioned on the bad side carries the signal to an earpiece in the good ear, but patients find BAHAs to be more effective and acceptable.
Disadvantages:
Cost. The cost of the BAHA and implantable kit is approximately £4000, and there are additional costs for hospital, surgeon and anaesthetists fees. Some insurance companies cover the costs fully, others cover only the cost of surgery, while some do not cover any of the costs. There may be additional ongoing costs for repairs, treatment of minor skin infections etc.
Surgery is required to implant the device, although this can be under local or general anaesthetic, and can usually be carried out as a single stage procedure. As the middle ear is not entered, the risks of middle ear surgery such as damage to hearing or the facial nerve do not apply.
Minor skin infections can occur around the abutment.
Trauma may damage or loosen the fixture, particularly in children, and replacement may be required.
BAHAs are a safe and effective treatment for moderate conductive and mixed losses.
Middle Ear Implants:
Middle ear implants are a recent innovation and can be totally or partially implantable, but the only device available in the UK at present is the Vibrant Soundbridge, a partially implanted device. A transducer is surgically attached to the ossicles in the middle ear. Sound is detected by a microphone in the audio processor, digitised and the signal transferred to a receiver surgically implanted beneath the skin. The signal is then transmitted to the transducer which vibrates, moving the ossicles and transferring energy to the cochlea.
.Figure 2. Middle Ear Implant
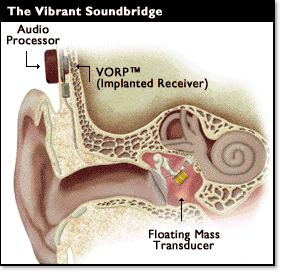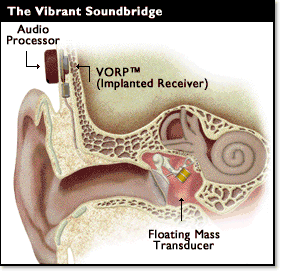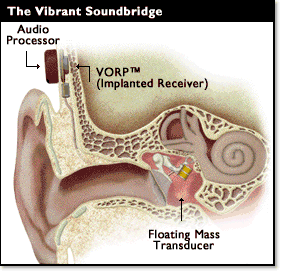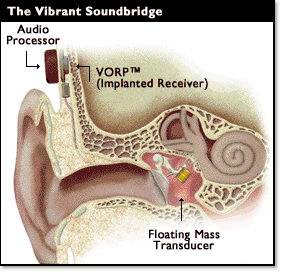
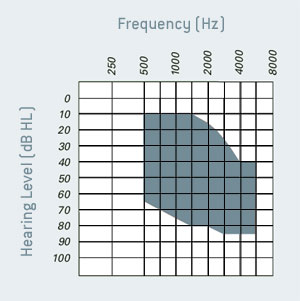
Indications: These devices are suitable for patients aged 18 and above with mild to severe sensorineural hearing losses as shown below.
Some patients cannot wear hearing aids due to infections, abnormal ear canals or inability to tolerate a hearing aid, and for these a middle ear implant may be a solution.
Advantages: Why should someone want a middle ear implant as opposed to a conventional aid?
There is less feedback and whistling with these devices, allowing greater amplification.
They do not block the ear canal so patients get less echoing of their own voice.
Because the ear canal is not blocked, it is less likely to become sweaty, moist and infected.
It is claimed that sounds seem clearer and more natural than with a conventional hearing aid.
They are more comfortable, and of course the cosmetic appearance is also a factor.
Over 1000 people worldwide have been implanted with the Soundbridge device and the vast majority continue to use it.
Extended applications: More recently, middle ear implants have been used for mixed and conductive losses and novel approaches such as round window positioning have been used with promising results.
Disadvantages:
The cost of implanting these devices is undoubtedly greater than the cost of a hearing aid.
MRI scanning of the head and neck is contra-indicated with the implant in situ.
There are always risks associated with surgery, including anaesthetic risks and specific risks associated with middle ear surgery.
There is concern about potential long term damage to the ossicles or hearing, as constant movement against the ossicles may cause erosion of the ossicles, and non-physiological stimulation may cause inner ear damage. These devices have only been used for the last 5 years so long term data is not available, but the information available so far suggests that this not a significant problem.
Cochlear Implants:
Cochlear implants are used to improve hearing for patients with severe to profound sensorineural hearing loss (thresholds greater than 70dB). In most cases of sensorineural loss inner ear hair cells are damaged while the bipolar neurones in the centre of the cochlea are relatively preserved. These can be stimulated by an electrode in the cochlear duct to send signals along the auditory nerve. As they are arranged tonotopically, stimulation at different sites along the cochlear duct and in different patterns can stimulate distinct populations of bipolar neurones and hence code for pitch, while intensity coding is determined by stimulation current. The basic design is shown below.
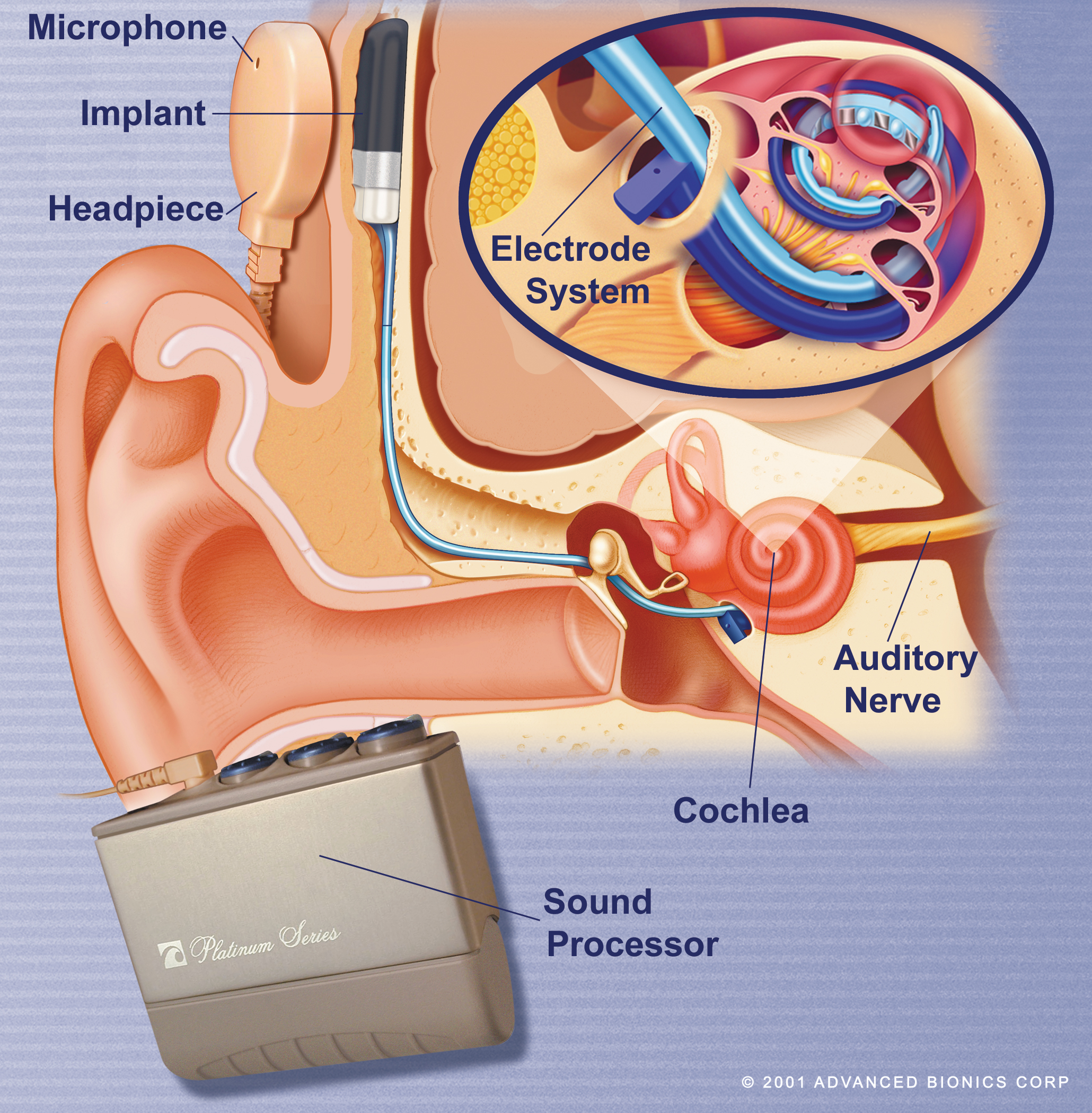 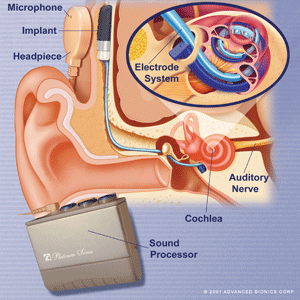
A receiver/stimulator package is implanted subcutaneously, with the electrode being inserted into the cochlea. Sound is detected by the microphone and digitized by the processor. Electrical signals are transferred across the scalp skin via the headpiece to the receiver and then along the electrode to stimulate bipolar neurones in the cochlea.
Modern devices incorporate the microphone and processor in a small behind the ear device similar to a hearing aid.

Patients undergo a thorough medical and audiological evaluation to determine whether an implant is likely to help. Surgery takes 2 hours and is usually performed under general anaesthetic. Patients usually go home the next day, and the device is “switched on” after 4 to 6 weeks. Rehabilitation and adjustment of the device programmes (“mapping”) is an ongoing process. It is important to realize the limitations of a cochlear implant. It does not restore normal hearing, music appreciation is limited, and a great deal of time and effort is required for rehabilitation. However, deaf children can develop normal speech and language, and deaf adults can have a conversation over the telephone with a stranger, and these results were unthinkable 30 years ago.
Indications:
Anyone who will do better with a cochlear implant than the best possible hearing aids is a candidate. As the results from implants improve, the range of suitable candidates continues to increase. Current guidelines are:
patients with unaided thresholds of 70dB or worse
aided thresholds of 50dB or worse
poor speech recognition e.g. sentence scores less than 50%,
and such patients should be referred for formal assessment by a cochlear implant unit.
Which patients are suitable?
Post-lingually deafened adults and children, i.e. those that have acquired speech and language and then become deaf do very well with a cochlear implant.
Pre-lingually deafened children e.g. those born deaf or deafened before acquiring speech and language can do very well if implanted in time. The central nervous system pathways for speech and language are laid down in the first few years of life, but this plasticity is diminishes after the age of 5. Children implanted before this age can do well, and the best results are obtained if children are implanted before the age of 2. After the age of 5 results decline, and implanting a congenitally deaf teenager produces poor results.
The auditory nerve must be intact on the side to be implanted. Where there is no auditory nerve, a brainstem implant may help.
The cochlea should be patent for the best results. In some cases e.g. after meningitis the cochlear duct is quickly obliterated by new bone or fibrous tissue making it impossible to insert an electrode fully. Alternative modified electrodes and techniques can be employed and the results may not be as good as with a standard electrode but are still worthwhile. Congenitally abnormal cochleas may also require modifications to the standard implantation technique.
Which patients are not suitable?
Patients with a unilateral hearing loss.
Patients with a mild or moderate loss will do better with a hearing aid.
Pre-lingually deafened adults.
Patients who cannot learn to use the implant.
Patients unfit for surgery.
Patients with unrealistic expectations.
Disadvantages:
Cost. The device itself costs approximately £18,000, and the total cost of assessment, device, surgery and postoperative care for a year in the NHS is approximately £40,000- but these are patients who are not benefiting from the strongest hearing aids and for whom there is no alternative.
There are risks associated with surgery, including the risks of anaesthesia and the risks of middle ear surgery. Postoperative infections and device failure can also occur.
There was concern recently of an increased risk of meningitis following insertion of a cochlear implant. Investigations revealed a slight increase in risk related to the design of one particular device, and this has now been withdrawn. There is probably a very slightly increased risk of meningitis after cochlear implantation with any device, but this is minimized by patients receiving a pneumococcal vaccine prior to surgery, and antibiotics are used to cover the surgery. These precautions are now part of the routine for cochlear implant surgery.
The Future:
Ongoing improvements in hardware and software design produce improved results year on year. Current software research is focused on improving speech perception in the presence of background noise and on improving music perception.
Bilateral cochlear implants are now established and provide improved localization of sound and improved speech perception in the presence of background noise. Cost utility analysis however suggests that the increased cost outweighs the increased benefit in a resource limited system such as the NHS. Recent NICE guidelines (2009) accept that bilateral implants are of significant benefit for children and should be offered to all suitable children. However, for adults NICE guidelines suggest unilateral implants are more cost effective. Some patients opt to pay for the second implant themselves and are in general happy with the increased benefit they receive.
Many patients have a sloping hearing loss with some preservation of low frequency hearing. Hybrid devices which combine short electrodes to stimulate high frequency neurones with acoustic amplification to amplify low frequencies have been implanted and the early results are very promising. Modified “soft surgery” techniques are used to minimize loss of residual hearing at the time of surgery. These devices are not suitable for patients with a progressive hearing loss.
Totally implantable devices are being developed, but a few problems such as battery design and positioning of the microphone are yet to be resolved. Such devices may be available commercially within the next few years.
Brainstem Implants:
In some cases, the auditory nerve may be damaged, for example due to trauma or previous surgery. In such cases, stimulation in the cochlea will be of no benefit as the signals cannot be transmitted to the brain. Auditory brainstem implants cater for this situation by placing a flat electrode on the surface of the brainstem overlying the cochlear nuclei. There is a degree of tonotopic organization of the neurones which can be used for preferential frequency stimulation, but this is not as easy to use as in the cochlea. The results from brainstem implants are very limited in comparison to cochlear implants, with few patients developing speech discrimination, but the ABI can provide some auditory inflammation to aid with lipreading.
Conclusions:
Patients with mild to moderate conductive or mixed loss may be helped by a BAHA, particularly where chronic ear discharge makes wearing a conventional aid impossible.
Patients with mild to severe sensorineural or mixed loss who cannot or will not wear a conventional hearing aid may benefit from a middle ear implant.
Patients with severe to profound sensorineural loss who do not benefit from hearing aids may benefit from a cochlear implant, and in children early implantation gives the best results.
Mr. Azhar Shaida
Consultant ENT Surgeon
The Royal National Throat Nose and Ear Hospital, London
The Harley Street ENT Clinic, 109 Harley Street, London
Mr. Shaida has a special interest in Otology and Cochlear Implants, and can be contacted on 02079151616 (Secretary) or via email at doctor@ent-doctor.co.uk.
|

