|
Cochlear implants have evolved significantly from the early single channel implants in the 1960’s to the multichannel implants available today. They have gone from being a research tool to an accepted mainstream treatment for severe to profound sensorineural hearing loss. They are now used for children, used bilaterally, and increasingly used as hybrid devices with hearing preservation techniques for treatment of sloping sensorineural hearing losses. Wider applications such as treatment of single sided deafness and tinnitus are being explored.
Cochlear implants work on the principle that in most cases of sensorineural hearing loss the inner ear hair cells are damaged while the bipolar neurones in the centre of the cochlea are relatively preserved. These can be stimulated by an electrode in the cochlear duct to send signals along the auditory nerve. As the bipolar neurones are arranged tonotopically along the modiolus, stimulation at different sites along the cochlear duct and in different patterns can stimulate distinct populations of bipolar neurones and hence code for pitch, while intensity coding is determined by stimulation current. The basic design is shown below.
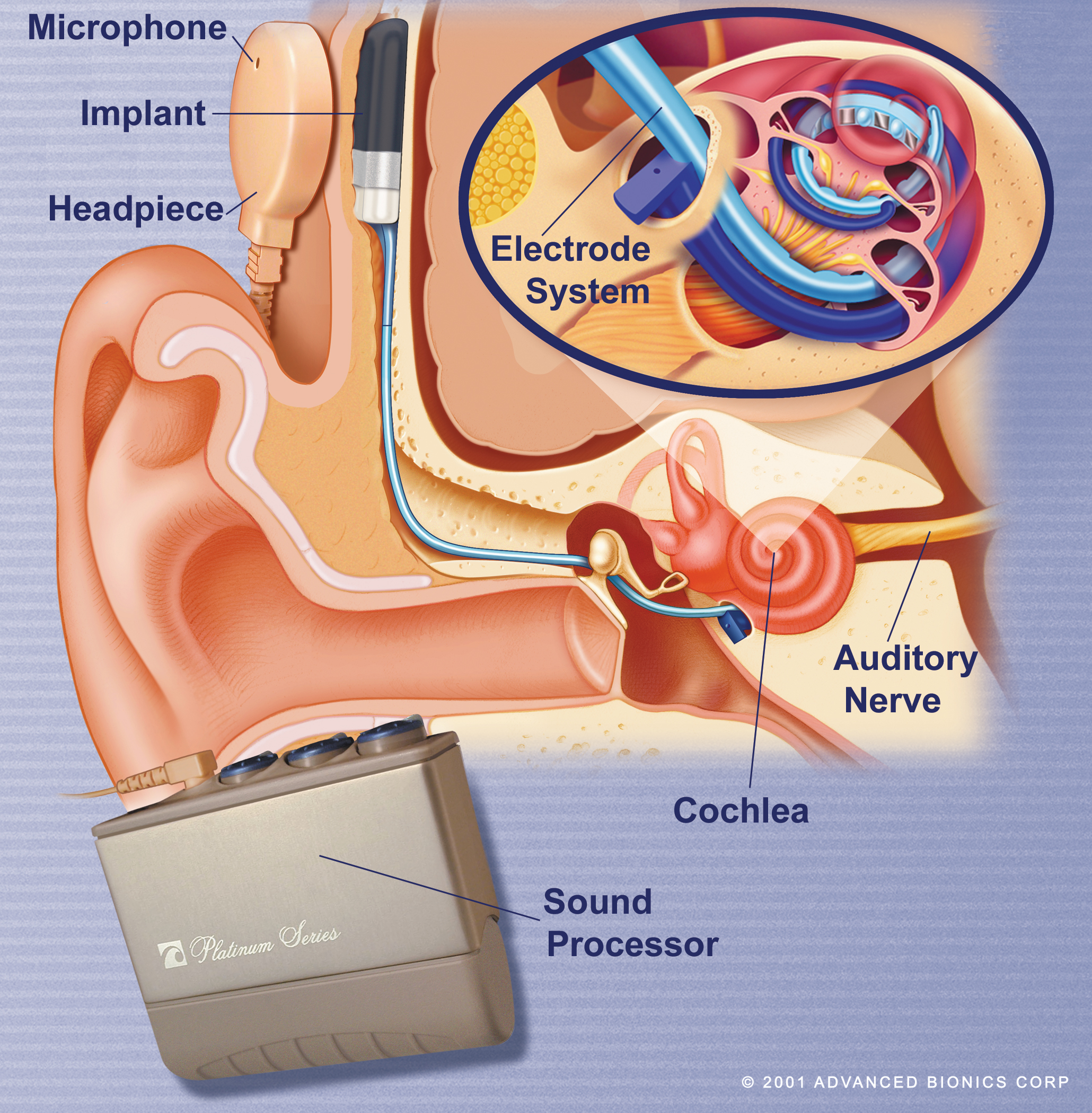 
A receiver/stimulator package is implanted subcutaneously, with the electrode being inserted into the cochlea. Sound is detected by the microphone and digitized by the processor. Electrical signals are transferred across the scalp skin via the headpiece to the receiver and then along the electrode to stimulate bipolar neurones in the cochlea.
Modern devices incorporate the microphone and processor in a small behind the ear device similar to a hearing aid. The coil is placed over the implanted receiver and held in position by a magnet.

Patients undergo a thorough medical and audiological evaluation to determine whether an implant is likely to help. Surgery takes 2 hours and is usually performed under general anaesthetic. Patients usually go home the next day, and the device is “switched on” after 4 to 6 weeks. Some patients report hearing whistles and beeps when the device is switched on, while some report hearing metallic artificial speech straight away. For most patients it takes time to get used to the implant and to be able to interpret the strange electronic sounds as speech, and it can take a year or even two to get maximum benefit from the implant. Rehabilitation and adjustment of the device programmes (“mapping”) is an ongoing process, with regular weekly sessions fro the first six weeks and then further sessions depending on requirements.
It is important to realize the limitations of a cochlear implant. It does not restore normal hearing, music appreciation is limited, and a great deal of time and effort is required for rehabilitation. However, children born deaf can develop normal speech and language, and many deaf adults can go back to having a conversation over the telephone with a stranger. These results were unthinkable 30 years ago.
Indications:
Anyone who will do better with a cochlear implant than the best possible hearing aids is a candidate. As the results from implants improve, the range of suitable candidates continues to increase. Current guidelines are:
Patients with unaided thresholds of 90dB or worse at 4kHz and 8kHz (UK NICE guidelines, although in other countries thresholds worse than 70dB are considered).
Poor speech recognition e.g. BKB or IHR sentence scores less than 50% (although in other countries scores up to 70% are considered).
Aided thresholds of 50dB or worse are also an indicator that the patient may benefit from a cochlear implant.
Such patients should be referred for formal assessment by a cochlear implant unit.
Which patients are suitable?
Post-lingually deafened adults and children, i.e. those that have acquired speech and language and then become deaf, do very well with a cochlear implant. In these patients the auditory pathways from ear to speech and hearing areas of the brain have been established, and can be stimulated again with a cochlear implant.
Pre-lingually deafened children e.g. those born deaf or deafened before acquiring speech and language can do very well if implanted in time. The central nervous system pathways for speech and language are laid down in the first few years of life, but this plasticity is diminishes after the age of 5. Children implanted before this age can do well, and the best results are obtained if children are implanted before the age of 2. After the age of 5 results decline, and implanting a congenitally deaf teenager produces poor results.
The auditory nerve must be intact on the side to be implanted. Where there is no auditory nerve, a brainstem implant may help.
The cochlea should be patent for the best results. In some cases e.g. after meningitis the cochlear duct is quickly obliterated by new bone or fibrous tissue making it impossible to insert an electrode fully. Alternative modified electrodes and techniques can be employed and the results may not be as good as with a standard electrode but are still worthwhile. Congenitally abnormal cochleas may also require modifications to the standard implantation technique.
Which patients are not suitable?
Patients with a unilateral hearing loss.
Patients with a mild or moderate loss will do better with a hearing aid.
Pre-lingually deafened adults (although increasing numbers of early deafened late implanted or “non-traditional” candidates are being implanted, with good subjective benefit and moderate objective hearing benefit).
Patients who cannot learn to use the implant.
Patients unfit for surgery.
Patients with unrealistic expectations.
Disadvantages:
Cost. The device itself costs approximately £18,000, and the total cost of assessment, device, surgery and postoperative care for a year in the NHS is approximately £40,000- but these are patients who are not benefiting from the strongest hearing aids and for whom there is no alternative.
There are risks associated with surgery, including the risks of anaesthesia and the risks of middle ear surgery. Postoperative infections and device failure can also occur.
There was concern a few years ago of an increased risk of meningitis following insertion of a cochlear implant. Investigations revealed a slight increase in risk related to the design of one particular device, and this has now been withdrawn. There is probably a very slightly increased risk of meningitis after cochlear implantation with any device, but this is minimized by patients receiving a pneumococcal vaccine prior to surgery, and antibiotics are used to cover the surgery. These precautions are now part of the routine for cochlear implant surgery.
Available Devices:
Devices are available in the UK from Cochlear, Advanced Bionics, MedEl and Neurelec. In the past, each manufacturer had devices suitable for certain inner ear malformations, cochlear obliteration, fibrosis etc. so the choice of device might be determined by surgical and radiological considerations, Now most manufacturers produce a variety of devices suitable for a variety of situations, but there may still be situations for example where a stiffer electrode is preferred and this may guide device selection.
In terms of outcomes, manufacturers all claim their device is the best, and conflicting studies are available, but it appears that at present, despite variations in hardware and speech processing, the hearing outcomes are similar with all devices.
As only single cochlear implants are offered by the NHS to adult patients in the UK, some patients chose to opt for a Neurelec binaural implant which is funded by the NHS and costs the same as a single implant, but offers two electrodes, one for each ear, providing stimulation for both ears.
The Future:
Ongoing improvements in hardware and software design produce improved results year on year. Current software research is focused on improving speech perception in the presence of background noise and on improving music perception.
Bilateral cochlear implants are now established and provide improved localization of sound and improved speech perception in the presence of background noise. Cost utility analysis however suggests that the increased cost outweighs the increased benefit in a resource limited system such as the NHS. Recent NICE guidelines (2009) accept that bilateral implants are of significant benefit for children and should be offered to all suitable children. However, for adults NICE guidelines suggest the additional cost of a second implant is not justified by the additional benefit it provides. Some patients opt to pay for the second implant themselves and are in general happy with the increased benefit they receive.
Many patients have a sloping hearing loss with some preservation of low frequency hearing. Hybrid devices which combine short electrodes to stimulate high frequency neurones with acoustic amplification to amplify low frequencies have been implanted and the early results are very promising. Modified “soft surgery” techniques are used to minimize loss of residual hearing at the time of surgery. These devices are not suitable for patients with a progressive hearing loss.
Totally implantable devices are being developed, but a few problems such as battery design and positioning of the microphone are yet to be resolved. Such devices may be available commercially within the next few years.
|

